 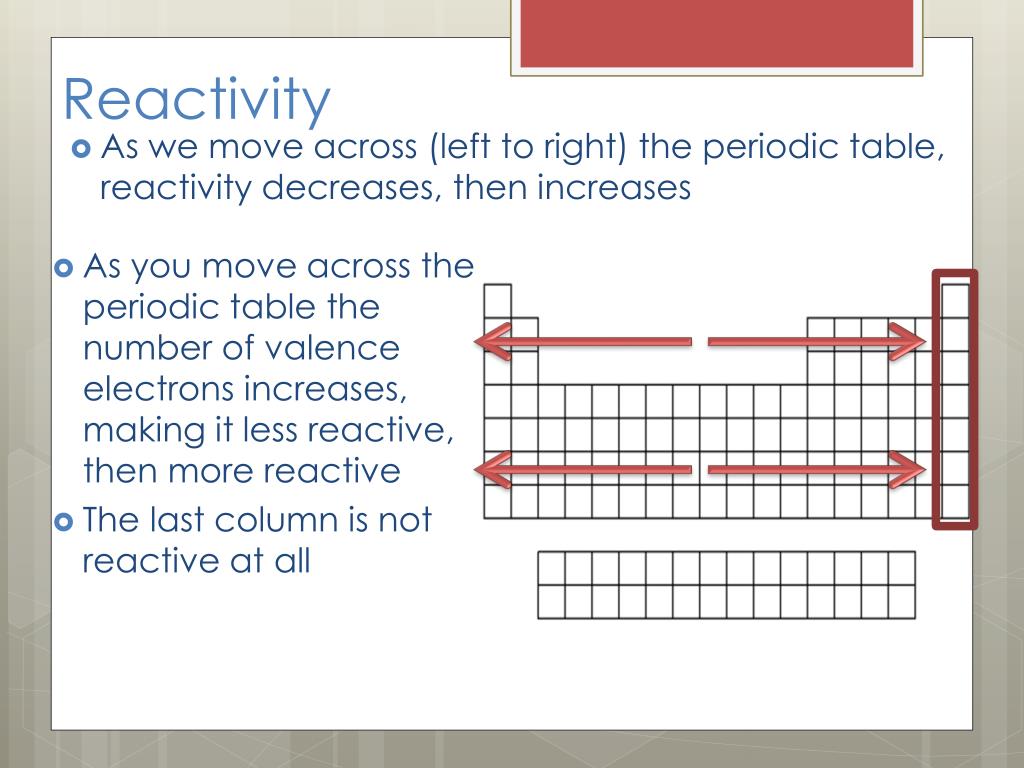  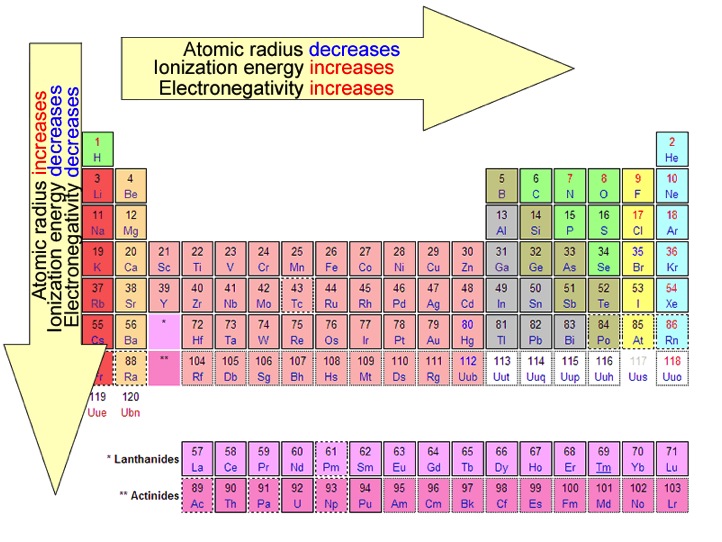
Whilst there are some trends that run along the rows, the most important trends are found when you go down the groups. reactivity of the alkali metals with water. The patterns in the periodic table are usually referred to as trends because the patterns are not exact and in some cases, you can't measure them directly e.g. These have 'trivial names' - the alkali metals, the halogens and the noble gases. You need to know a little about several of these groups, particularly groups 1, 7 and 0. These properties can easily be explained now that we know the atomic structure - metallic bonding, with its 'sea of delocalised electrons' explains most of them.Īll of the scientists who contributed to the development of the periodic table arranged groups of similar elements in vertical columns. Metals all have similar basic properties, for example they conduct heat and electricity, they are malleable and ductile, they are shiny when freshly cut and so on. We can see that the metals appear on the left hand side of the table whilst the much smaller group of non-metals is to the right. The biggest pattern is perhaps concerning the original classification of the elements into metals and non-metals by French scientist Antoine de Lavoisier. Mendeleev's clever insight meant that definite patterns could be seen, patterns that now we take for granted. When they did this they discovered that the order of elements corresponded to Mendeleev's table. When protons were discovered, scientists worked out the atomic number of each element and arranged them in atomic number order. When the first 'missing' element was discovered, it fitted the pattern that Mendeleev had predicted, which was good evidence that he had been on the right track. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
